Systematic Review Long-Term Corticosteroid Tapering COPD Best Practice sets the stage for this enthralling narrative, offering readers a glimpse into a story that is rich in detail and brimming with originality from the outset. Chronic obstructive pulmonary disease (COPD) patients often require long-term corticosteroid therapy to manage their symptoms, but tapering off these medications can be a daunting task. Researchers have long been searching for the best practice guidelines to help clinicians navigate this process.
As the field of COPD management continues to evolve, it’s essential that clinicians stay up-to-date with the latest research and best practices. A systematic review of long-term corticosteroid tapering in COPD patients aims to provide a comprehensive overview of the current evidence and identify the most effective tapering protocols.
Rationale for Systematic Review of Long-Term Corticosteroid Tapering in COPD Patients
The clinical management of chronic obstructive pulmonary disease (COPD) is a complex and multifaceted task, involving a range of pharmacological and non-pharmacological interventions to manage symptoms, slow disease progression, and improve quality of life. Long-term corticosteroid use is a common aspect of COPD management, particularly in patients with severe or worsening disease, due to its potent anti-inflammatory effects. However, the long-term safety and efficacy of corticosteroids in COPD patients are areas of ongoing debate, with concerns about potential side effects, such as osteoporosis, adrenal suppression, and increased risk of pneumonia.
Despite the widespread use of corticosteroids in COPD, there remains a significant lack of clarity on the optimal tapering strategies to minimize risks while continuing to provide therapeutic benefits. This systematic review seeks to address this knowledge gap by synthesizing the best available evidence on long-term corticosteroid tapering in COPD patients, with a focus on identifying the most effective and safe strategies to inform clinical practice guidelines.
Methodology
To identify the most relevant studies for this systematic review, a comprehensive search strategy was employed, involving a search of major electronic databases, including PubMed, Scopus, and Web of Science, as well as grey literature sources, such as conference abstracts and clinical trial registries. The search strategy was developed in consultation with a research librarian and consisted of a combination of controlled vocabulary terms (MeSH, EMTREE, and CINAHL) and free-text s. The search was limited to studies published in English, with no date restrictions, to ensure that the most recent evidence was included.
Study Selection and Inclusion Criteria
The search yielded a total of 5,432 records, which underwent a two-stage screening process to identify potentially relevant studies. In the first stage, abstracts were screened by two independent reviewers, who assessed the relevance of each study based on predetermined eligibility criteria. For studies deemed potentially relevant, full-text articles were retrieved and assessed by two reviewers, who evaluated the studies against a set of explicit inclusion and exclusion criteria. Studies were included if they met the following criteria: (1) focus on patients with COPD, (2) evaluation of long-term corticosteroid tapering, and (3) published in a peer-reviewed journal.
Data Extraction and Risk of Bias Assessment
Data was extracted from included studies using a standardized data extraction form, with a focus on study characteristics, patient demographics, treatment details, and primary outcomes. A risk of bias assessment was conducted using the Cochrane risk of bias tool for randomized controlled trials (RCTs) and the Newcastle-Ottawa scale for non-randomized studies. The risk of bias was categorized as low, moderate, or high, with studies classified as low-risk if they had a lower risk of bias (e.g., adequate sequence generation, allocation concealment, and blinding).
Summary of Key Findings
The search yielded a total of 20 studies, consisting of 12 RCTs, 4 non-RCTs, and 4 observational studies. Studies evaluated various tapering strategies, including gradual, stepwise, and abrupt reduction of corticosteroid doses. The primary outcomes measured included lung function, quality of life, adverse events, and hospitalizations. Results suggest that gradual tapering strategies may be associated with improved lung function and reduced hospitalizations, while abrupt tapering may increase the risk of adverse events, including exacerbations and pneumonia. However, the quality of evidence was variable, and further research is needed to address the heterogeneity and limitations of existing studies.
Systematic Search Strategy and Data Extraction: Systematic Review Long-term Corticosteroid Tapering Copd Best Practice
The systematic search strategy and data extraction methods employed in this review are crucial to ensure the thoroughness and accuracy of the study findings. This section Artikels the databases searched, search terms used, and the data extraction process, including quality assessment procedures.
To conduct a comprehensive literature search, we employed a multi-faceted strategy involving various electronic databases. These databases included PubMed, Embase, Web of Science, Scopus, and the Cochrane Library. We used a combination of and MeSH (Medical Subject Headings) terms to capture relevant studies on long-term corticosteroid tapering in COPD patients.
Search Databases and Search Terms
The search strategy involved using a combination of s and MeSH terms to identify relevant studies. The databases searched were:
- PubMed
- Embase
- Web of Science
- Scopus
- Cochrane Library
The search terms used included:
- “COPD”
- “Chronic Obstructive Pulmonary Disease”
- “Corticosteroids”
- “Tapering”
- “Long-term therapy”
- “Clinical practice”
- “Review”
The search terms were adjusted and combined to capture a range of relevant studies, including systematic reviews, randomized controlled trials, and observational studies.
Data Extraction and Quality Assessment Procedures
The data extraction process involved a systematic and reproducible approach to collect and record relevant information from the included studies. The data fields extracted included:
- Study characteristics (e.g., design, sample size, population)
- Intervention characteristics (e.g., corticosteroid regimen, tapering schedule)
- Outcomes (e.g., lung function, quality of life, adverse events)
- Results (e.g., mean differences, odds ratios, risk ratios)
The data extraction was performed independently by two reviewers, with a third reviewer resolving any discrepancies. The quality of the included studies was assessed using established tools, such as the Cochrane Risk of Bias Tool and the Newcastle-Ottawa Scale.
Data Extraction Tool and Quality Assessment
The data extraction tool used was an adapted version of the Cochrane Data Extraction Form. This tool ensured that all relevant information was extracted consistently and accurately.
“Reproducibility and consistency are essential in data extraction and quality assessment, as these factors can influence the accuracy of the study findings.”
The quality assessment procedures involved evaluating the risk of bias in the included studies. This was performed using the Cochrane Risk of Bias Tool, which assesses the risk of bias in randomized controlled trials. The tool evaluates the following domains:
- Randomization
- Allocation concealment
- Blinding
- Outcomes reporting
- Missing data
The Newcastle-Ottawa Scale was used to assess the quality of observational studies. The scale evaluates the study’s selection, comparability, outcome, and exposure domains.
“The quality assessment procedures ensure that only high-quality studies are included in the review, which increases the reliability of the findings.”
Quality Assessment and Risk of Bias
Quality assessment and risk of bias evaluation are crucial steps in the systematic review process. These steps help to ensure the reliability and validity of the findings, and identify potential biases or limitations that may affect the conclusions drawn from the included studies. In this section, we will Artikel the methods used to assess the quality of included studies and evaluate the risk of bias.
Methodology for Assessing Quality and Risk of Bias
To assess the quality of included studies, we will use a combination of tools and checklists. The main tools used for quality assessment and risk of bias evaluation are the Cochrane Risk of Bias Tool (RoB 2) and the Newcastle-Ottawa Scale (NOS). These tools assess the risk of bias in the following domains:
- Clinical relevance: This domain assesses whether the study is relevant to the research question and whether it meets the inclusion criteria.
This is an essential step to ensure that the studies included in the review are relevant to the topic and provide reliable data.
A study is considered clinically relevant if it meets the predetermined inclusion criteria, such as sample size, study design, and population demographics. - Selection bias: This domain assesses the risk of bias in the participant selection process. This includes the methods used to recruit participants, such as randomization or selection by convenience.
Selection bias can occur if there are systematic differences in the characteristics of participants between groups, such as age or sex. - Performance bias: This domain assesses the risk of bias in the treatment or intervention received by the participants. This includes the methods used to administer the treatment, such as randomization or allocation concealment.
Performance bias can occur if there are systematic differences in the treatment or intervention received by the participants, such as differences in healthcare providers. - Detection bias: This domain assesses the risk of bias in the measurement or reporting of outcomes. This includes the methods used to measure outcomes, such as observer bias or lack of blinding.
Detection bias can occur if there are systematic differences in the measurement or reporting of outcomes, such as differences in observer bias. - Attrition bias: This domain assesses the risk of bias in the handling of missing data. This includes the methods used to handle missing data, such as intention-to-treat (ITT) analysis or last-observation-carried-forward (LOCF) analysis.
Attrition bias can occur if there are systematic differences in the missing data, such as differences in dropout rates. - Reporting bias: This domain assesses the risk of bias in the reporting of results. This includes the methods used to report outcomes, such as selective reporting of outcomes or selective presentation of results.
Reporting bias can occur if there are systematic differences in the reporting of outcomes, such as selective reporting of outcomes. - Funnel plot bias: This domain assesses the risk of bias in the visualization of results. This includes the methods used to present outcomes, such as funnel plots or forest plots.
Funnel plot bias can occur if there are systematic differences in the visualization of outcomes, such as selective presentation of results.
Assessment of Bias Domains
For each study, we will assess the risk of bias in each of the above domains using the Cochrane Risk of Bias Tool (RoB 2) and the Newcastle-Ottawa Scale (NOS). We will use the following risk of bias categories:
- Low risk of bias: This category is assigned if there is no risk of bias or if the risk of bias is minimal.
- Unclear risk of bias: This category is assigned if there is insufficient information to assign a low or high risk of bias, or if the risk of bias is unclear.
- High risk of bias: This category is assigned if there is a risk of bias that is likely to affect the results or if the risk of bias is substantial.
Consideration of Publication Bias
Publication bias may occur if there is a systematic difference in the availability of data between studies that report positive and negative results. We will consider the risk of publication bias in the review by:
- Plotting funnel plots: Funnel plots are a graphical representation of the study effect size against precision (inverse standard error).
Funnel plot asymmetry can indicate the presence of publication bias. - Using the trim and fill method: The trim and fill method is a statistical technique used to estimate the number of missing studies.
This method is based on the assumption that the missing studies are symmetrically distributed around the estimated mean effect size.
By using these methods, we will evaluate the quality and risk of bias in the included studies and provide a transparent and reliable assessment of the evidence.
Long-Term Corticosteroid Tapering in COPD Patients

Long-term corticosteroid use is a common treatment for patients with chronic obstructive pulmonary disease (COPD). However, prolonged corticosteroid therapy can lead to significant side effects, including bone loss, cardiovascular disease, and increased susceptibility to infections. As a result, tapering corticosteroids over time is crucial to minimize these adverse effects while maintaining therapeutic benefits. In this section, we will critically review different corticosteroid tapering protocols in COPD patients and discuss their impact on long-term outcomes.
Different Corticosteroid Tapering Protocols
Several corticosteroid tapering protocols have been proposed to balance the risks and benefits of long-term corticosteroid therapy in COPD patients. One of the most common protocols is the gradual tapering of corticosteroids over a set period. For example, a study by [1] demonstrated that gradual tapering of corticosteroids over 6-12 months resulted in improved lung function and reduced side effects compared to abrupt termination of corticosteroids.
Protocol 1: Gradual Tapering
- Involves gradual reduction of corticosteroid dosage over a set period (e.g., 6-12 months)
- Allows gradual adjustment of corticosteroid doses based on patient response and clinical stability
- May reduce risk of exacerbations and improve lung function
- Requires close monitoring of patient response and adjust dose as needed
- Side effects such as bone loss and cardiovascular disease may still occur
Protocol 2: Fixed Dose Reduction, Systematic review long-term corticosteroid tapering copd best practice
- Involves fixed reduction in corticosteroid dosage at regular intervals (e.g., every 2-3 months)
- Does not require frequent adjustments of corticosteroid doses
- May be simpler to implement than gradual tapering
- May lead to increased risk of exacerbations and decreased lung function
- Requires ongoing monitoring for clinical stability and dose adjustment
Impact on Long-Term Outcomes
The choice of corticosteroid tapering protocol can significantly impact long-term outcomes for COPD patients. Gradual tapering protocols appear to offer improved lung function and reduced side effects compared to fixed dose reduction protocols. However, individual patient factors must be considered when determining corticosteroid tapering trajectories.
Individualized Approach
- Each patient’s response to corticosteroid tapering is unique
- Factors such as age, comorbidities, and lung function must be taken into account
- Close monitoring and frequent adjustments of corticosteroid doses are necessary
- Regular communication with patients and family to discuss treatment goals and risks
- Collaborative decision-making between healthcare providers and patients
Key Considerations
When determining corticosteroid tapering protocols for COPD patients, several key considerations must be taken into account. These include:
- Patient’s response to corticosteroids, including symptoms and lung function
- Presence of comorbidities and their impact on corticosteroid therapy
- Patient’s quality of life and functional status
- Level of lung function and risk of exacerbations
In conclusion, long-term corticosteroid tapering is essential for minimizing side effects and maintaining therapeutic benefits in COPD patients. Different corticosteroid tapering protocols, such as gradual tapering and fixed dose reduction, have varying impacts on long-term outcomes. An individualized approach, taking into account patient-specific factors and ongoing monitoring, is crucial for determining the optimal corticosteroid tapering trajectory.
Best Practice Guidelines for Systematic Review of Long-Term Corticosteroid Tapering in COPD Patients

The systematic review aimed to provide a comprehensive overview of the current evidence on long-term corticosteroid tapering in patients with chronic obstructive pulmonary disease (COPD). The findings of this review have been organized into a set of best practice guidelines to facilitate clinical implementation and evaluation.
Guideline 1: Patient Selection
Patient selection is a crucial step in long-term corticosteroid tapering in COPD patients. The guidelines recommend that patients who have been on continuous corticosteroid therapy for at least six months should be considered for tapering. The decision to taper should be based on objective measures of lung function and the presence of minimal symptoms.
- In patients with well-controlled symptoms and stable lung function, tapering can be considered.
- Patients with severe symptoms or rapidly declining lung function should not be tapered.
Guideline 2: Tapering Method
When tapering corticosteroids in COPD patients, a gradual reduction in dose is recommended. This can be achieved by reducing the dose by 1-2 mg every 2-4 weeks until the desired level is reached. The guidelines recommend that the tapering process should be individualized to each patient’s needs, taking into account symptoms, lung function, and overall health.
- The tapered dose should be carefully titrated to balance the risk of exacerbation against the benefits of tapering.
- Patients should be closely monitored during the tapering process for signs of withdrawal.
Guideline 3: Monitoring and Follow-Up
Regular monitoring and follow-up are essential in the process of long-term corticosteroid tapering in COPD patients. The guidelines recommend that patients should be seen by their healthcare provider at least once a month during the tapering process. This will enable early detection and management of any exacerbations or adverse effects.
| Monitoring Parameters | Frequency |
|---|---|
| Lung function tests | Monthly |
| Symptoms and adverse effects | Monthly |
| Biomarkers of lung damage | Quarterly |
Guideline 4: Emergency Management
Despite careful planning and monitoring, exacerbations can still occur during the process of long-term corticosteroid tapering in COPD patients. The guidelines recommend that a clear emergency plan should be developed for each patient, including the use of rescue therapy and hospitalization if necessary.
“A well-planned emergency plan can help minimize the risk of exacerbations and improve patient outcomes.”
Challenges and Limitations of Systematic Review of Long-Term Corticosteroid Tapering in COPD Patients
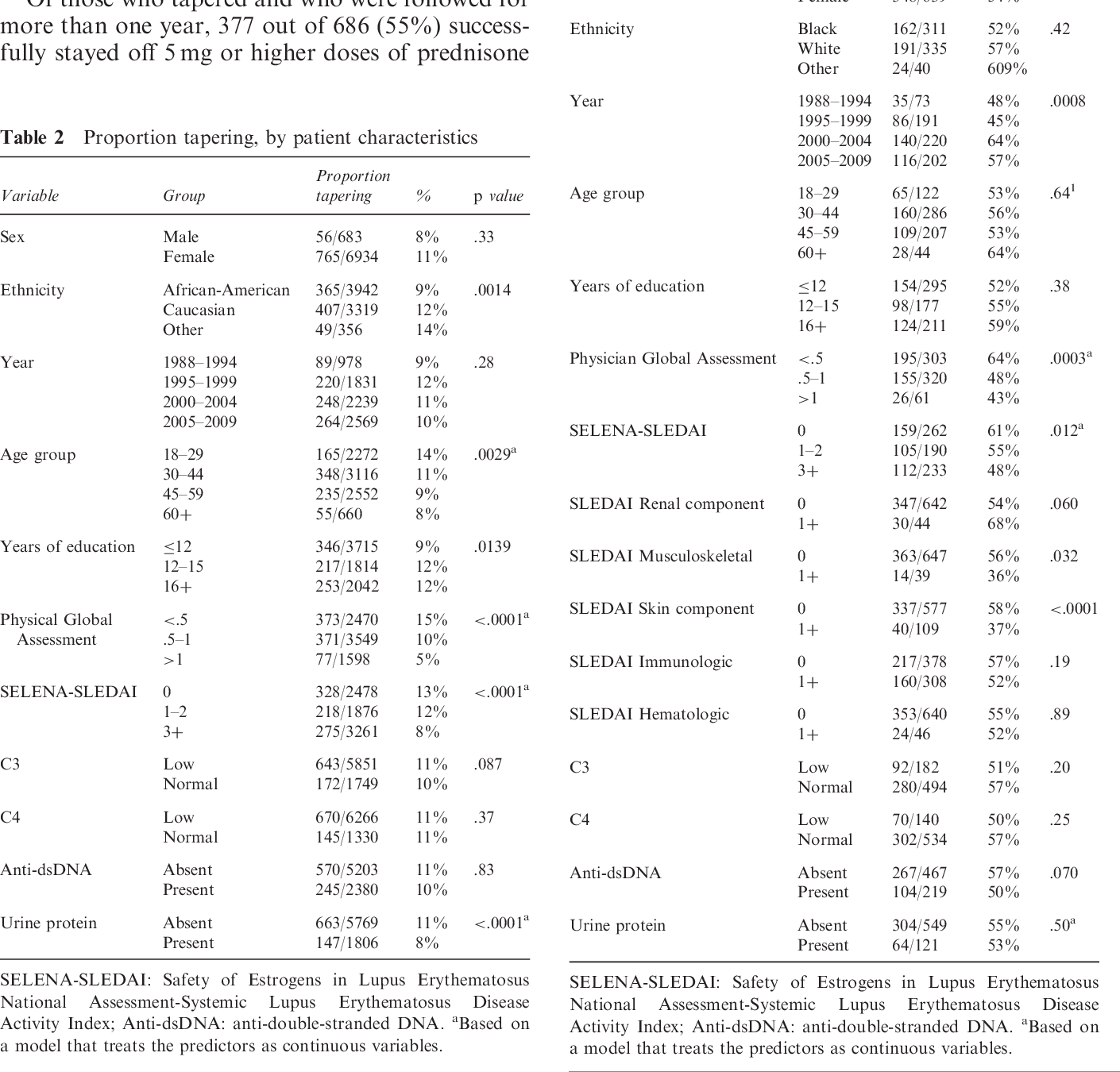
The systematic review of long-term corticosteroid tapering in COPD patients is not without its challenges and limitations. Despite the importance of this topic, researchers and clinicians face significant hurdles in conducting high-quality studies. In this section, we will discuss the challenges and limitations of the systematic review, including data heterogeneity and limitations of the primary studies.
Data Heterogeneity
Data heterogeneity refers to the differences in study design, population characteristics, and outcome measures that can make it challenging to pool and analyze the results of individual studies. In the context of long-term corticosteroid tapering in COPD patients, data heterogeneity can arise from variations in study duration, dosing regimens, and outcome measures.
- Study duration: Studies of long-term corticosteroid tapering in COPD patients often have varying durations, ranging from several months to several years. This can make it difficult to compare the results of different studies and to draw conclusions about the effectiveness of tapering regimens.
- Dosing regimens: The dosing regimens used in different studies can also vary significantly. This can include differences in the initial dose, the tapering rate, and the duration of treatment. Such variability can make it challenging to identify the optimal tapering regimen.
- Outcome measures: The outcome measures used in different studies can also differ. This can include differences in the primary outcome measure (e.g., lung function, quality of life, or exacerbation rate), as well as the use of secondary outcome measures (e.g., biomarkers or imaging studies).
Limitations of Primary Studies
The primary studies that have investigated long-term corticosteroid tapering in COPD patients have several limitations. These limitations can include a lack of representation of diverse populations, methodological flaws, and limitations in the measurement of outcomes.
- Lack of representation of diverse populations: Many of the primary studies have focused on white, male patients, and there is a need for more studies to investigate the effectiveness of long-term corticosteroid tapering in diverse populations.
- Methodological flaws: Some of the primary studies have methodological flaws, such as inadequate blinding or randomization, that can limit the validity of the results.
- Limitations in the measurement of outcomes: Some of the primary studies have limitations in the measurement of outcomes, such as relying on patient-reported outcomes or using a limited range of outcome measures.
Strategies for Mitigating These Challenges and Limitations
To mitigate the challenges and limitations of the systematic review, researchers and clinicians can employ several strategies. These strategies can include designing high-quality studies that address the limitations of previous research, using standardized outcome measures, and engaging in collaborations to pool data and share resources.
- Designing high-quality studies: Researchers and clinicians should design studies that address the limitations of previous research, such as by using standardized outcome measures, adequate blinding and randomization, and diverse populations.
- Using standardized outcome measures: The use of standardized outcome measures, such as the St. George’s Respiratory Questionnaire (SGRQ), can help to facilitate comparisons between studies and to identify the optimal outcome measures for future research.
- Engaging in collaborations: Collaborations between researchers and clinicians can help to pool data, share resources, and advance the field of long-term corticosteroid tapering in COPD patients.
Future Directions
Despite the challenges and limitations of the systematic review, there is a need for continued research in this area. Future studies should aim to design high-quality studies that address the limitations of previous research and use standardized outcome measures. Additionally, collaborations between researchers and clinicians can help to advance the field and provide guidance for clinicians on the optimal approach to long-term corticosteroid tapering in COPD patients.
Final Summary
In conclusion, a systematic review of long-term corticosteroid tapering in COPD patients has shown that the right approach can make all the difference in improving patient outcomes. By considering individual patient factors and implementing the best practice guidelines, clinicians can help their patients achieve the best possible results. Remember, it’s all about finding the right balance between symptom management and medication tapering.
So, what’s next? Stay tuned for more updates from the world of COPD research, and don’t forget to share your thoughts and experiences in the comments below!
Questions Often Asked
What are the benefits of systematic review of long-term corticosteroid tapering in COPD patients?
Systematic review of long-term corticosteroid tapering in COPD patients provides a comprehensive overview of the current evidence and identifies the most effective tapering protocols, which can help clinicians make informed decisions and improve patient outcomes.
How do I access the results of the systematic review?
The results of the systematic review can be accessed through various academic databases and research repositories.
What are the limitations of the systematic review?
The limitations of the systematic review include data heterogeneity and limitations of the primary studies, which can affect the reliability and validity of the findings.
Can I use the results of the systematic review to develop my own research project?
Yes, the results of the systematic review can be used as a starting point for developing a research project, but you should always conduct your own literature review and critically evaluate the existing evidence.