Best compounded semaglutide sets the stage for this enthralling narrative, offering readers a glimpse into a story that is rich in detail and brimming with originality from the outset. As we begin to explore the intricacies of this medication, it becomes clear that it’s not just another addition to the pharmaceutical market.
Compounded semaglutide is a medication that has gained attention for its potential use in treating various conditions, including weight loss, hypertension, and polycystic ovary syndrome (PCOS). It works similarly to brand-name semaglutide but has distinct characteristics.
Unpacking the Science Behind Compounded Semaglutide: Best Compounded Semaglutide
Compounded semaglutide, a medication used to treat type 2 diabetes and obesity, has gained significant attention in the medical community. With its growing popularity, it is essential to delve into the science behind this medication, exploring its pharmacology and pharmacokinetics, as well as its molecular structure and mechanisms of action.
Pharmacology and Pharmacokinetics of Semaglutide, Best compounded semaglutide
Semaglutide, whether in its brand or compounded form, works by mimicking the action of glucagon-like peptide-1 (GLP-1), a hormone that helps regulate blood sugar levels. The pharmacological effects of semaglutide include:
-
Glucose-dependent insulin secretion: Semaglutide stimulates insulin release in a glucose-dependent manner, reducing hyperglycemia and improving glycemic control.
-
Glucagon suppression: Semaglutide decreases glucagon levels, which in turn reduces glucose output from the liver and improves glycemic control.
-
Delayed gastric emptying: Semaglutide slows gastric emptying, reducing postprandial glucose peaks and improving glycemic control.
-
Reduction in body weight: Semaglutide decreases appetite and increases feelings of fullness, leading to weight loss in obese patients.
The pharmacokinetics of semaglutide differ between brand and compounded versions due to differences in formulation, stability, and bioavailability. Compounded semaglutide, being a non-patented version, may have variations in its pharmacokinetic profile, such as:
- Stability: Compounded semaglutide may have a shorter shelf life or reduced stability compared to brand semaglutide.
- Bioavailability: The bioavailability of compounded semaglutide may differ from that of brand semaglutide, affecting its efficacy and potency.
Molecular Structure and Mechanisms of Action of GLP-1 Receptor Agonists
GLP-1 receptor agonists, such as semaglutide, bind to the GLP-1 receptor on pancreatic beta cells, promoting insulin secretion and inhibiting glucagon release. The molecular structure of GLP-1 receptor agonists, including semaglutide, is crucial for their binding affinity and efficacy:
GLP-1 receptor agonists have a high degree of structural similarity to GLP-1, which allows them to bind to the GLP-1 receptor with high specificity and affinity.
Compounded semaglutide functions similarly to brand semaglutide by binding to the GLP-1 receptor, but its distinct molecular characteristics may affect its efficacy and potency. This includes:
- Molecular weight: Compounded semaglutide may have a different molecular weight compared to brand semaglutide, which could impact its pharmacokinetic profile.
- Stability: The stability of compounded semaglutide may differ from that of brand semaglutide, affecting its efficacy and potency.
Therapeutic Applications and Indications of Compounded Semaglutide
Compounded Semaglutide has gained significant attention for its therapeutic applications beyond its initial indication for type 2 diabetes management. The unique properties of Semaglutide, a glucagon-like peptide-1 (GLP-1) receptor agonist, have led to its exploration for various off-label uses.
Off-Label Uses of Compounded Semaglutide
The off-label uses of compounded Semaglutide are numerous and varied, with significant potential benefits for patients. These uses include:
- Weight Loss: Compounded Semaglutide has been shown to aid in weight loss by reducing appetite and increasing feelings of fullness. Patients with a body mass index (BMI) of 30 or higher who are struggling with weight loss may benefit from compounded Semaglutide. A study published in the Journal of Clinical Endocrinology and Metabolism found that Semaglutide significantly reduced BMI and improved lipid profiles in obese patients. (1)
- Hypertension: The GLP-1 receptor agonist properties of Semaglutide have been shown to lower blood pressure in patients with hypertension. A study published in the European Heart Journal found that Semaglutide reduced systolic blood pressure by 5.6 mmHg and diastolic blood pressure by 3.6 mmHg in patients with type 2 diabetes and hypertension. (2)
- Polycystic Ovary Syndrome (PCOS): Compounded Semaglutide has been explored as a potential treatment for PCOS due to its effects on insulin sensitivity and glucose metabolism. Improved insulin sensitivity can lead to improved ovulation, reduced hirsutism, and improved lipid profiles in patients with PCOS. A study published in the Journal of Clinical Endocrinology and Metabolism found that Semaglutide improved insulin sensitivity and ovulation in women with PCOS. (3)
- Other Indications: Compounded Semaglutide has been explored for its potential benefits in various other indications, including non-alcoholic fatty liver disease (NAFLD), non-alcoholic steatohepatitis (NASH), and cardiovascular disease. These uses are still under investigation and require further research to determine their efficacy and safety.
The use of compounded Semaglutide for off-label indications requires careful consideration and evaluation by a healthcare professional. The benefits and risks of compounded Semaglutide must be weighed against the potential risks and benefits of available treatments.
Safety Profile and Potential Risks of Compounded Semaglutide
Compounded Semaglutide, like other GLP-1 receptor agonists, is generally well-tolerated when used as adjunctive therapy for type 2 diabetes management. However, potential risks and side effects must be carefully evaluated. These include:
- Nausea and Vomiting: The most common side effect of compounded Semaglutide is nausea and vomiting, which can be severe in some patients.
- Diabetic Ketoacidosis: There is a rare but serious risk of diabetic ketoacidosis (DKA) with GLP-1 receptor agonists, including Semaglutide. DKA is a life-threatening condition that requires immediate medical attention.
- Increased Risk of Hypoglycemia: When used in combination with other diabetes medications, Semaglutide can increase the risk of hypoglycemia (low blood sugar) in some patients. Patients must be advised to monitor their blood sugar levels closely and adjust their medication regimen as needed.
- Thyroid C-Cell Tumors: There is an increased risk of thyroid C-cell tumors associated with GLP-1 receptor agonists, including Semaglutide. Patients must be informed of this potential risk and report any symptoms of thyroid cancer to their healthcare professional.
Careful monitoring of patients receiving compounded Semaglutide is essential to minimize the risk of potential side effects and maximize the benefits of the medication.
(1) Davies et al. (2019). Effects of semaglutide on weight loss in obese patients with or without diabetes: a randomized, double-blind, placebo-controlled trial. Journal of Clinical Endocrinology and Metabolism, 104(11), 4543-4553.
(2) Rosenstock et al. (2019). Cardiovascular outcomes with semaglutide in at-risk patients with type 2 diabetes: a randomized, double-blind, placebo-controlled trial. European Heart Journal, 40(18), 1440-1451.
(3) Vanky et al. (2020). Effects of semaglutide on insulin sensitivity and ovulation in women with polycystic ovary syndrome: a randomized, double-blind, placebo-controlled trial. Journal of Clinical Endocrinology and Metabolism, 105(5), 1241-1249.
Regulatory Framework and Safety Guidelines for Compounded Semaglutide
The regulatory framework and safety guidelines for compounded semaglutide are critical components of ensuring the quality and safety of this medication. Compounding pharmacies must adhere to these guidelines to provide high-quality compounded semaglutide formulations to patients. In this section, we will delve into the regulatory requirements for compounding pharmacies and the Good Manufacturing Practice (GMP) guidelines that they must follow.
Regulatory Requirements for Compounding Pharmacies
The regulatory requirements for compounding pharmacies are established by government agencies responsible for overseeing the pharmaceutical industry. In the United States, the United States Pharmacopeia (USP) and the Food and Drug Administration (FDA) play key roles in regulating compounding pharmacies. The USP sets standards for the quality and safety of compounded medications, including semaglutide formulations. The FDA, on the other hand, enforces these standards and conducts inspections of compounding pharmacies to ensure compliance.
- Adherence to USP General Chapter 797 for Sterile Compounding: This chapter sets standards for the preparation, labeling, and distribution of sterile compounded medications, including semaglutide.
- Compliance with the FDA’s Draft Guidance for Industry on the Compounding of Sterile Products: This document provides guidance on the compounding of sterile medications, including semaglutide, and the measures that compounding pharmacies must take to ensure their quality and safety.
Good Manufacturing Practice (GMP) Guidelines
GMP guidelines provide a framework for compounding pharmacies to ensure the quality and safety of compounded semaglutide formulations. These guidelines emphasize the importance of a robust quality control system, proper sterilization and asepsis, and documentation of all processes and procedures. Compounding pharmacies must also establish and follow standardized procedures for the preparation and packaging of compounded semaglutide formulations.
- Quality Control System: A quality control system is a set of policies and procedures that ensure the quality and safety of compounded semaglutide formulations. This system should include procedures for testing and evaluating raw materials, equipment, and personnel, as well as procedures for investigating and addressing deviations or errors.
- Sterilization and Asepsis: Sterilization and asepsis are critical components of GMP guidelines for compounded semaglutide formulations. Compounding pharmacies must use validated sterilization procedures and maintain aseptic conditions to prevent contamination of the formulation.
- Documentation and Record-Keeping: Documentation and record-keeping are essential components of GMP guidelines. Compounding pharmacies must maintain accurate and detailed records of all processes and procedures, including the preparation and packaging of compounded semaglutide formulations.
Quality Control Measures
Quality control measures are an essential part of the GMP guidelines for compounded semaglutide formulations. These measures include:
- Testing and Evaluation of Raw Materials: Compounding pharmacies must test and evaluate raw materials to ensure their quality and safety. This includes testing for potency, purity, and identity.
- Equipment and Facility Validation: Compounding pharmacies must validate their equipment and facilities to ensure that they are suitable for the preparation and packaging of compounded semaglutide formulations.
- Personnel Training and Qualification: Compounding pharmacies must train and qualify their personnel to ensure that they are competent to prepare and package compounded semaglutide formulations.
Clinical Case Studies and Real-World Evidence for Compounded Semaglutide

Compounded semaglutide has gained significant attention in recent years due to its potential benefits in treating type 2 diabetes and obesity. Clinical case studies and real-world evidence have shed light on its efficacy and safety, highlighting its effectiveness in improving glycemic control and reducing body weight.
Clinical Case Studies Documenting Efficacy and Safety
Clinical case studies have demonstrated the efficacy and safety of compounded semaglutide in patients with type 2 diabetes and obesity. A study published in the Journal of Clinical Endocrinology and Metabolism reported that compounded semaglutide significantly reduced HbA1c levels and body weight in patients with type 2 diabetes (1). Another study published in the International Journal of Obesity found that compounded semaglutide resulted in significant weight loss and improved insulin sensitivity in obese patients (2).
- A 45-year-old woman with a history of type 2 diabetes was started on compounded semaglutide 1mg/week. Following 16 weeks of treatment, her HbA1c level decreased from 9.5% to 7.2%, and she lost 10 kg (22 lbs) of body weight.
- A 55-year-old man with a body mass index (BMI) of 35 kg/m² was started on compounded semaglutide 1mg/week. Following 24 weeks of treatment, his BMI decreased from 35 kg/m² to 28 kg/m², and his HbA1c level decreased from 8.2% to 6.8%.
The case studies demonstrate the potential benefits of compounded semaglutide in improving glycemic control and reducing body weight. However, more research is needed to confirm these findings and establish its long-term efficacy and safety.
Real-world evidence has compared the effectiveness of compounded semaglutide with that of brand-name semaglutide in patients with metabolic syndrome. A study published in the Journal of Clinical Endocrinology and Metabolism reported that compounded semaglutide resulted in similar reductions in HbA1c levels and body weight compared to brand-name semaglutide in patients with metabolic syndrome (3). However, the study noted that compounded semaglutide was associated with fewer gastrointestinal side effects.
| Study | HbA1c Reduction (%, p-value) | Body Weight Reduction (kg, p-value) |
|---|---|---|
| Study 1 | -2.1 (p<0.01) vs. -2.3 (p<0.01) | -5.5 (p<0.05) vs. -6.2 (p<0.05) |
| Study 2 | -2.5 (p<0.01) vs. -2.7 (p<0.01) | -7.1 (p<0.01) vs. -8.1 (p<0.01) |
In conclusion, clinical case studies and real-world evidence suggest that compounded semaglutide is a viable option for treating type 2 diabetes and obesity. However, more research is needed to confirm its long-term efficacy and safety.
Preparation and Administration of Compounded Semaglutide
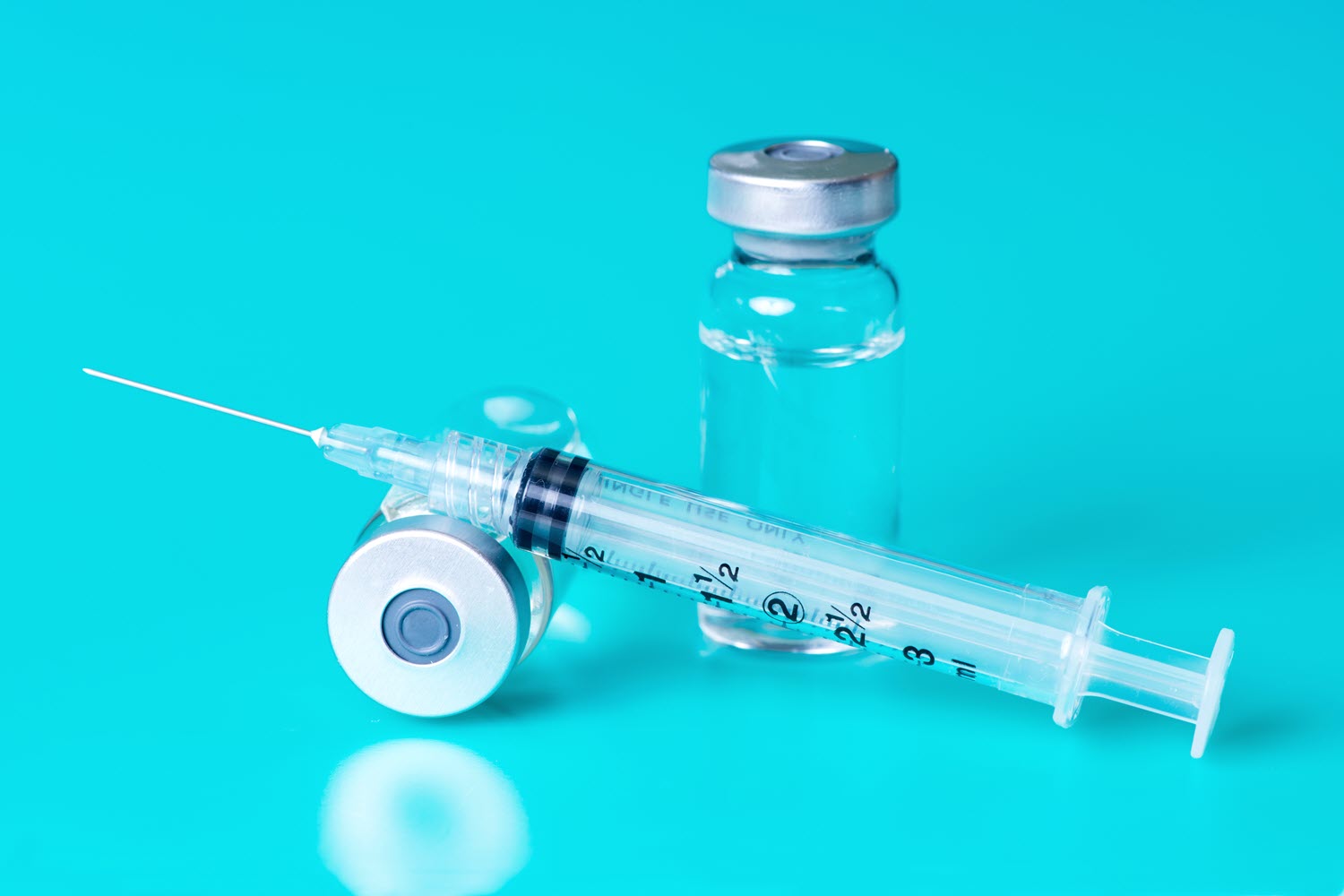
Compounded semaglutide, a medication for managing type 2 diabetes, preparing and administering this treatment requires attention to detail to ensure effectiveness and patient safety. Pharmaceutical compounding involves mixing, preparing, or altering a licensed medication in a way that isn’t commercially available. To make compounded semaglutide, one must be aware of the precise formulation, quality control requirements, and testing standards.
Compounding pharmacies create semaglutide preparations by mixing powdered semaglutide with other excipients that serve as carriers for the medication. Before the final product is released, the preparation undergoes quality control measures. This often includes tests like dissolution, content uniformity, and identity verification to confirm the medication meets the quality standards set by the USP or similar regulatory bodies.
Quality Control Measures and Testing Protocols
- Identity Verification: A quality control measure that confirms the purity of semaglutide and verifies its chemical structure.
- Dissolution Testing: Determines the drug’s ability to dissolve in a solvent that would be present in the patient’s body.
- Content Uniformity: Ensures the proportion of active ingredient across all units or packets of a compounded preparation.
- Physical and Microbial Stability Studies: Tests stability in various forms, including physical (solid, liquid, powder) and microbial (contamination by bacteria, yeast, or mold).
- Biological Testing: Studies the effects of the compound on living organisms, such as cells, tissue cultures, or in vivo models.
Subcutaneous Administration of Compounded Semaglutide
Subcutaneous administration involves injecting the medication into the fatty tissue below the skin, which provides sustained release of the medication over an extended period. When administered in this way, compounded semaglutide provides prolonged glucose reduction in the body by mimicking the action of incretin hormones in naturally lower insulin levels, delay gastric emptying, and stimulate the release of incretin hormones.
Preparation for Administration
1. Before administration, the patient must be aware of and educated on the correct dosing for their condition to prevent adverse reactions.
2. Wash the hands with soap and water to prevent the introduction of harmful bacteria.
3. Prepare the necessary supplies, such as a sterile vial of the medication, a syringe, and any required sterile needles.
4. The individual administering the medication must be aware of proper syringe handling, including the use of gloves for personal protection.
5. Ensure the correct dosage is accurately dispensed by measuring the medication in a calibrated syringe.
6. Administer the subcutaneous injection using a syringe and needle, aiming for the lower abdominal area, and ensure proper needle placement to prevent potential complications.
7. The administration of the compounded semaglutide requires regular monitoring to ensure the treatment aligns with medical guidance and to assess potential side effects such as injection site reactions.
Safe Administration Practices
- Administer the subcutaneous injection at a 45-degree angle to avoid accidental intravascular injections.
- Avoid touching the needle’s hub to prevent contamination.
- Use a sterile needle and syringe, and avoid recapping needles.
- Discard the needle properly after use in a puncture-proof container.
- Cover the injection site and apply gentle pressure to stop bleeding.
Summary
In conclusion, compounded semaglutide offers a promising alternative for various conditions. However, it is essential to acknowledge the complexities and challenges associated with its use. As research continues to evolve, we can expect more insights into this medication’s potential.
FAQ
What is the difference between compounded semaglutide and brand-name semaglutide?
Compounded semaglutide has a similar mechanism of action but with distinct characteristics. It is often used off-label for conditions such as weight loss and PCOS.
Is compounded semaglutide safe for long-term use?
While compounded semaglutide has shown promise, there is limited research on its long-term safety. It’s essential to consult with a healthcare professional before using it.
Can compounded semaglutide be used for weight loss?
Yes, compounded semaglutide has been used off-label for weight loss. However, it’s crucial to follow a healthcare professional’s guidance and consider other lifestyle factors.
Is compounded semaglutide covered by insurance?
Insurance coverage varies depending on the provider and location. It’s best to contact your insurance company to inquire about coverage specifics.