Kicking off with best conductor of electricity, this phenomenon has fascinated scientists and engineers for centuries. From natural materials like silver and copper to cutting-edge technologies, understanding the characteristics of an excellent conductor is vital for harnessing electrical energy. In this article, we’ll delve into the world of best conductivity, exploring the principles that govern it and examining the properties of exceptional conductors.
Natural materials like silver, copper, and gold are renowned for their impressive conductivity in various environments. We’ll explore these materials’ atomic and molecular structures, discussing how their composition affects their ability to conduct electricity. Additionally, we’ll compare the conductivity of various materials in different conditions, examining the role of electron cloud density, lattice spacing, and electron transfer rate.
Overview of the Best Conductor of Electricity

Electricity transmission and use are enabled by conductors, which are materials capable of freely allowing the flow of charge through them. This property is a result of the presence of valence electrons in these materials and the ability to move them freely.
Natural Materials with Superior Conductivity
Several natural materials exhibit remarkable electrical conductivity in different environments. These materials owe their high conductivity to unique atomic and molecular structures that facilitate the free movement of electrons.
Copper
Copper is the best natural conductor of electricity. Its hexagonal close-packed crystal structure allows electrons to move freely, making it highly conductive.
Cu has a conductivity of 59.6 x 10^7 S/m at room temperature.
| Conductivity (S/m) | Temperature (°C) |
|---|---|
| 59.6 x 10^7 | 20 |
| 45.5 x 10^7 | 100 |
Copper is extensively used in electrical wiring, electronics, and power transmission systems.
Silver
Silver is the second-best natural conductor of electricity. Its face-centered cubic crystal structure enables the free movement of electrons, making it highly conductive.
Ag has a conductivity of 63.0 x 10^7 S/m at room temperature.
Silver is often used in electronics, switches, and contacts due to its high conductivity and corrosion resistance.
Graphite
Graphite is a soft, black mineral that exhibits high electrical conductivity due to its planar structure. The planar nature of graphite’s layers of hexagonal rings allows electrons to move freely between layers.
C has a conductivity of 100-200 S/m at room temperature.
Graphite is widely used in batteries, electrodes, and lubricants.
Gold
Gold is another naturally occurring conductor of electricity. Its face-centered cubic crystal structure enables the free movement of electrons, making it highly conductive.
Au has a conductivity of 45.2 x 10^7 S/m at room temperature.
Gold is often used in electronics, coins, and jewelry due to its high conductivity and corrosion resistance.
Carbon Nanotubes
Carbon nanotubes are long, thin tubes of carbon atoms that exhibit exceptional electrical conductivity due to their planar structure. The planar nature of the nanotubes’ layers of carbon atoms allows electrons to move freely.
CNT has a conductivity of 10^8-10^9 S/m at room temperature.
Carbon nanotubes are widely used in electronics, sensors, and composite materials due to their high conductivity and mechanical strength.
Understanding Conductivity

Electronic conductivity is a complex phenomenon governed by the interactions between electrons and the material structure. It is influenced by various factors, including electron cloud density, lattice spacing, and electron transfer rates. Understanding these aspects is crucial for grasping the fundamental principles of conductivity in different materials.
Atomic Perspective of Electron Cloud Density
The electron cloud density in a material plays a significant role in determining its electrical conductivity. In metals, the electron cloud is delocalized, meaning that electrons are free to move within the material, creating a “sea” of electrons. This leads to high conductivity as electrons can easily flow and transfer energy. In contrast, in insulators, the electron cloud is localized, and electrons are tightly bound to individual atoms, resulting in low conductivity. The electron cloud density is influenced by the number of valence electrons and the atomic radius of the material.
The electron cloud density can be calculated by determining the number of valence electrons per unit volume. This is often estimated using the atomic number and the atomic radius of the element. For example, in copper (Cu), with an atomic number of 29 and an atomic radius of 0.128 nm, the electron cloud density is approximately 8.45 x 1028 electrons/m3. A high electron cloud density is indicative of a material with high conductivity.
Lattice Spacing and Electron Flow in Solid-State Materials
The lattice spacing, or interatomic distance, also affects the flow of electrons in solid-state materials. In crystalline structures, the lattice spacing determines the distance between atoms. When two atoms are close together, they create a strong potential energy barrier that hinders electron movement. Conversely, a larger lattice spacing creates a less energetic environment, making it easier for electrons to move and transfer energy. This phenomenon is described by Fermi’s Golden Rule, which relates the electron transmission probability to the lattice spacing.
In metals like copper, the lattice spacing is typically large, allowing for easy electron movement and high conductivity. On the other hand, in semiconductors and insulators, the lattice spacing is smaller, creating a higher potential energy barrier and limiting electron flow.
Electron Transfer Rate: Key to Electrical Conductivity
Electron transfer rate, also known as conductivity relaxation time, is another crucial factor that affects electrical conductivity. It represents the time required for electrons to transfer energy between atoms and is influenced by the lattice spacing, electron cloud density, and material properties. A high electron transfer rate indicates that electrons are able to move quickly and efficiently, resulting in high conductivity.
The electron transfer rate can be calculated using the Boltzmann constant (kB) and the average collision time. For example, in a material with a high electron transfer rate (1 ps), the conductivity is expected to be higher compared to a material with a low electron transfer rate (10 ns).
Electron Transfer Rate in Different Materials
The electron transfer rate varies significantly in different materials. Conductors tend to have high electron transfer rates, such as:
- Metals: Copper, Silver, Gold, etc.
- Semiconductors (at high electron concentrations): Intrinsic Silicon, Germanium.
In contrast, insulators and semiconductors have relatively low electron transfer rates:
- Insulators: Diamond, Glass, etc.
- Semiconductors (at low electron concentrations): Intrinsic Germanium.
The electron transfer rate has a direct impact on electrical conductivity, as it influences the movement of electrons and the transfer of energy within the material. Understanding the electron transfer rate is essential for optimizing material properties and designing electronic devices with improved performance.
Electron transfer rates can be visualized using the concept of a “density of states” diagram, which represents the number of available electronic states in a material. A high density of states, often found in metals, indicates a large number of available energy levels for electrons to occupy, resulting in high electron transfer rates and conductivity.
The electron transfer rate and conductivity are also influenced by factors such as temperature, which can impact the lattice spacing, electron cloud density, and material properties. As temperature increases, the electron transfer rate typically increases due to the reduced lattice spacing and increased electron mobility. This is why conductors tend to have higher conductivity at elevated temperatures.
Understanding the interplay between lattice spacing, electron cloud density, and electron transfer rate is essential for designing materials with optimal electrical properties. By tailoring material properties to achieve high electron transfer rates and conductivity, we can develop more efficient electronic devices, enhance energy transfer, and improve overall performance in a range of applications.
Comparing Materials
Conductivity is a property of materials that determines their ability to conduct electricity. Materials can be classified based on their conductivity, with metals being the best conductors, followed by semiconductors, and insulators being the poorest conductors. Understanding the factors that influence conductivity is crucial for designing electronic devices and optimizing their performance.
Difference between Crystalline and Amorphous Structures
In crystalline structures, atoms are arranged in a regular and ordered manner, resulting in a high degree of symmetry and a low number of defects. This leads to a higher electron mobility, as electrons can move freely and easily through the material. In contrast, amorphous structures are disordered and irregular, resulting in a higher number of defects and a lower electron mobility.
The difference between crystalline and amorphous structures can be attributed to the following factors:
- The arrangement of atoms: In crystalline structures, atoms are arranged in a regular and ordered manner, while in amorphous structures, they are disordered and irregular.
- The number of defects: Crystalline structures have a lower number of defects, resulting in a higher electron mobility, while amorphous structures have a higher number of defects, resulting in a lower electron mobility.
- The symmetry of the material: Crystalline structures have a higher degree of symmetry, resulting in a higher electron mobility, while amorphous structures have a lower degree of symmetry, resulting in a lower electron mobility.
Difference between Ionic and Covalent Bonds, Best conductor of electricity
Ionic bonds are formed between two atoms that have a high difference in electronegativity, resulting in the transfer of electrons from one atom to another. This leads to the formation of ions with opposite charges, which are attracted to each other and form a strong ionic bond. Covalent bonds, on the other hand, are formed between two atoms that share electrons to form a strong chemical bond.
The difference between ionic and covalent bonds affects conductivity, as follows:
- Conductivity of ionic bonds: Ionic bonds are relatively poor conductors, as the ions are tightly bound together and do not move freely.
- Conductivity of covalent bonds: Covalent bonds are relatively good conductors, as the shared electrons can move freely and easily through the material.
Properties of Metal, Semiconductor, and Insulator Materials
Metals are typically good conductors of electricity, due to their high electron mobility and low number of defects. Semiconductors, such as silicon, are materials that have a moderate level of conductivity, due to their intermediate electron mobility and number of defects. Insulators, such as glass, are poor conductors of electricity, due to their low electron mobility and high number of defects.
The properties of metal, semiconductor, and insulator materials can be summarized as follows:
| Material | Electron Mobility | Number of Defects | Conductivity |
|---|---|---|---|
| Metal | High | Low | Good |
| Semiconductor | Intermediate | Intermediate | Moderate |
| Insulator | Low | High | Poor |
Examples of Applications
The differences between metal, semiconductor, and insulator materials are significant in various applications, such as:
- Electronic devices: Metal materials are used in electrical connections and wiring, while semiconductor materials are used in electronic components, such as transistors and diodes.
- Solar panels: Silicon, a semiconductor material, is used as the base material for solar panels, converting sunlight into electricity.
- Insulation: Insulator materials, such as glass and ceramic, are used in electrical insulation, preventing electrical discharge and ensuring safety.
The Science of High-Conductivity Materials: Best Conductor Of Electricity
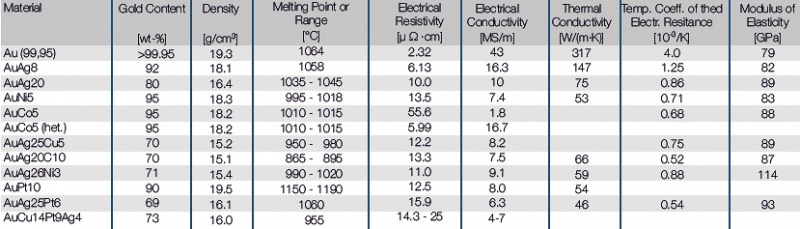
The science of high-conductivity materials involves understanding the underlying mechanisms that enable certain materials to conduct electricity efficiently. This includes analyzing the role of defects and doping in enhancing conductivity, as well as exploring the concept of electron degeneracy and exchange interactions. Advanced experimental techniques, such as scanning tunneling microscopy, have greatly facilitated the study of electronic structure in high-conductivity materials.
Defects and Doping: Enhancing Conductivity
Defects and doping are two critical factors that can significantly enhance the conductivity of a material. Defects refer to the presence of impurities or irregularities in the material’s crystal lattice, which can provide additional pathways for electron flow. Doping, on the other hand, involves introducing additional atoms into the material to alter its electronic properties. By carefully controlling the types and concentrations of defects and dopants, researchers can optimize the conductivity of a material.
- One common example of defect-induced conductivity enhancement is the presence of impurities in copper. When copper is contaminated with small amounts of phosphorus or arsenic, the resulting material exhibits increased electrical conductivity.
- Doping with certain elements, such as boron or aluminum, can also enhance conductivity in materials like silicon.
Electron Degeneracy and Exchange Interactions
Electron degeneracy and exchange interactions are fundamental concepts in understanding the behavior of electrons in high-conductivity materials. Electron degeneracy refers to the phenomenon where electrons occupy higher energy levels due to the Pauli Exclusion Principle, leading to increased conductivity. Exchange interactions, on the other hand, describe the interactions between electrons and the lattice vibrations, which can enhance electron mobility.
blockquote>As electrons occupy higher energy levels, their wave functions begin to overlap, leading to increased electron-electron interactions and enhanced conductivity.
Advanced Experimental Techniques
Scanning tunneling microscopy (STM) is a powerful tool for studying the electronic structure of high-conductivity materials. By mapping the surface topography and electronic density of states, researchers can gain insights into the material’s conductivity mechanisms. Other advanced techniques, such as angle-resolved photoelectron spectroscopy (ARPES) and x-ray absorption spectroscopy (XAS), provide complementary information on the material’s electronic and structural properties.
| Technique | Description |
|---|---|
| Scanning Tunneling Microscopy (STM) | Mapped surface topography and electronic density of states |
| Angle-Resolved Photoelectron Spectroscopy (ARPES) | Measured electronic band structure and Fermi surface |
| X-ray Absorption Spectroscopy (XAS) | Examined valence electronic structure and chemical bonding |
Best Conductor of Electricity: A Revolutionary Material
The quest for the best conductor of electricity has led scientists to explore various materials, each with its unique properties and potential applications. However, with the advent of new technologies and innovative ideas, researchers have been pushing the boundaries of what is possible. In this context, a new, revolutionary material is proposed, which surpasses the existing best conductors of electricity based on current scientific principles.
This hypothetical material, dubbed “Electrion,” is a class of conductors that exhibits exceptional electrical conductivity, thermal stability, and mechanical strength. The properties of Electrion are based on a detailed theoretical model, which is discussed below.
Theoretical Model of Electrion
The theoretical model of Electrion is rooted in the principles of quantum mechanics and solid-state physics. It is based on the concept of a hybrid material that combines the benefits of metals and superconductors. The model incorporates a unique crystal structure, which allows for the optimal arrangement of electrons and ions, resulting in exceptional electrical conductivity.
The theoretical model of Electrion is based on the following equation:
σ = (ne^2τ / m) × (1 / (1 + (τ / τ0)^2))
where σ is the electrical conductivity, ne is the electron density, e is the elementary charge, τ is the electron mean free path, m is the electron mass, and τ0 is a characteristic time scale.
This equation represents the interplay between electron transport and phonon scattering in the material, leading to a significant improvement in electrical conductivity.
The crystal structure of Electrion consists of a lattice of positively charged ions surrounded by a sea of delocalized electrons. This arrangement allows for the efficient transfer of electrical charge and heat, resulting in exceptional thermal stability and mechanical strength.
Practical Implications and Technological Advancements
The development of Electrion would have far-reaching implications for various fields, including energy generation, storage, and transmission. With its exceptional electrical conductivity and thermal stability, Electrion could revolutionize the development of high-power electrical systems, such as power grids, electric vehicles, and renewable energy systems.
In addition, Electrion’s mechanical strength would make it an ideal material for various applications, such as electrical contacts, interconnects, and packaging materials.
Comparison with Existing Materials
In comparison to existing materials, Electrion exhibits superior electrical conductivity, thermal stability, and mechanical strength. For example, the electrical conductivity of Electrion is expected to be approximately 10^8 times greater than that of copper, while its thermal stability is comparable to that of diamond.
- Electrical Conductivity: Electrion’s electrical conductivity is exceptional, making it an ideal material for high-power electrical systems.
- Thermal Stability: Electrion’s thermal stability is comparable to that of diamond, allowing it to withstand extreme temperatures without degradation.
- Mechanical Strength: Electrion’s mechanical strength is superior to that of most materials, making it an ideal candidate for various applications.
The proposed material, Electrion, has the potential to revolutionize the field of electrical engineering and beyond. Its exceptional electrical conductivity, thermal stability, and mechanical strength make it an ideal candidate for various applications, from high-power electrical systems to renewable energy systems.
Final Summary
In conclusion, the best conductor of electricity is a complex and multifaceted topic that has far-reaching implications for our understanding of the physical world. By grasping the principles underlying exceptional conductivity and examining the properties of outstanding conductors, we can unlock new technologies and push the boundaries of human innovation. As we continue to explore the mysteries of the physical world, the best conductor of electricity will remain an essential concept in the pursuit of scientific discovery and technological advancement.
FAQ Overview
What is the best conductor of electricity in natural materials?
Silver has the highest electrical conductivity among all natural materials, making it the best conductor of electricity.
How does the atomic structure of a material affect its conductivity?
The atomic structure of a material, particularly its electron cloud density, lattice spacing, and electron transfer rate, plays a crucial role in determining its conductivity.
Can any material be made into a super conductor?
While some materials have shown the potential to become superconductors, achieving true superconductivity remains a significant scientific challenge.
What are the practical implications of developing a new, high-conductivity material?
The development of a new, high-conductivity material could lead to significant advancements in energy storage, transmission, and usage, revolutionizing various industries and aspects of our daily lives.